The power of “p”: The p wave like a new predicting factor for Cardioembolic Stroke
Abbreviations Used:
Inter arterial blockage (IAB)
Incomplete Inter arterial blockage (IABi)
Complete Inter arterial blockage (IABc)
Ischemic stroke of undetermined source by incomplete study (ISUSi)
Electrocardiography (ECG)
Transient ischemic attack (TIA)
Computerized Tomography Scan (CTS)
Wide p wave (WPW).
Doppler Ultrasound of Supra-Aortic Trunks (DSAT)
Ischemic stroke (IS).
New Inter arterial block criteria (NIAB).
Magnetic Nuclear Resonance (MNR).
Transcranial Eco Doppler (TED).
Transthoracic echocardiogram (TE).
Transcranial eco doppler imaging tests (TED)
Abstract: the inter-atrial block (IAB) is a delay in atrial conduction defined as a wide p wave (WPW) on an electrocardiogram (ECG) recognized as a high specificity prediction factor for supraventricular tachycardia (SVA) prediction but weak for stroke prediction. New criteria for IAB (NIAB) appeared as the increase wide of p wave in the inferior leads, which has increased its predictive value. Objective to find a relationship between new NIAB and IS. Method: A retrospective study of 356 total patients: 178 cases admitted to the hospital for ISUSi (A group) and 178 controls admitted with no stroke history (B group) both groups had no prior arrhythmias and the same cardiovascular risks factors (cases and controls were matched1 to 1 by age, gender and CHA 2 DS 2 -vasc score). Defining the NIAB as: IABi: p bimodal ≥ 120 ms in I, I or III and IABc: p biphasic ≥ 120 ms in II, III and AVF. NIAB finding on the ECG was assessed in both groups at admission to the hospital. Data analysis was made to find IAB differences between groups in relation to age (<75; ≥75). Results: 356 patients (51% men; mean age 73,1+13,8; Barthel index 80,49+24,36 were included in study. WPW and NIAB findings were significantly more prevalent in group A (p≤0.000). Significant differences were found in age (<75; ≥75): stronger association WPW vs IABi and IABc in the younger group (OR 26,8(11,9-60,9) vs 21,5(7,27-63,5) and 25,43(3,3- 194) in contrast with a stronger association IABc and IABi vs WPW in the oldest group (OR 27,07(6,25-127,3) and 2,3(1,2-4,7) vs 9,6(4-23,2). Conclusions: WPW and NIAB were significantly related to ISUCi. Relevant differences were found considering age, being more prevalent WPW and IABi in younger and IABc in elder. Although more studies are needed, in the future these outcomes could justify primary prophylaxis with anticoagulation before SVA appears.
Introduction:
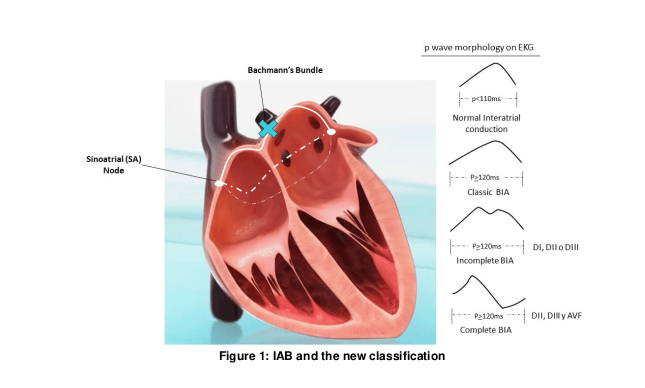
Atrial depolarization originates in Sinus Nodule. The stimulus after being generated is conducted by a specific route through Bachmann’s bundle, from top to bottom and from right to left direction, giving rise to the classical “p-wave” morphology in electrocardiogram (EKG), with a maximum length of 110 ms according to “The World Health Organization, the International Society and the Federation of Cardiology“. (1) Inter-atrial blockage (IAB) represents a dysfunction in the natural electrical conduction system between atrias (Bachmann’s bundle), causing a delay in its progression in early stages and a blockage in the advanced ones, forcing the use of alternatives electric channels (Ovallis fossa and Coronary sinus) to transmit stimulus from one atrium to the other, promoting an abnormal atrial depolarization (directed from bottom to top) and conditioning morphologic changes in “p-wave” (becoming wider and variegated). The IAB was first described in 1916(2) and classically defined as a slow inter-atrial depolarization, represented on the EKG as a wide “p wave” (> 110 ms). Object of multiple studies over the last 30 years, IAB has been proved as a predictor of supraventricular arrhythmias (Atrial Fibrillation and Flutter), as well as embolic cardiac events including ischemic strokes.
However, although classical studies proved IAB as an important 30-month predictor of supraventricular arrhythmias (3,4,5,6,7,8,9), its association with cardioembolic events has been disappointing, showing IAB as a predictor of low discriminative power. (10,11,12,13,14)
It was not until 2012 when a Spanish cardiologist, Dr. Bayes de Luna in his consensus entitled “A separate entity from left atrial enlargement” (3) , gave a dramatic turn to this situation by redefining and refining IAB’s diagnostic criteria, after analyzing over 80,000 electrocardiographic records. According to Bayes de
Luna, the IAB is characterized by a slow conduction through Bachmann’s bundle and should be represented on the EKG with a “p-wave” wider than classically defined (≥120ms instead of >110ms) and given that this conduction is done through two alternatives routes with different trajectories (Ovallis fossa and Coronary sinus), “p-wave” should have a morphology different from the classical one, which could only be observed in specific EKG leaves. In this way Bayes de Luna proposes an IAB subdivision depending on degree of dysfunction and conduction route used (Figure 1):
• Incomplete IAB or first grade blockage: defined as a bimodal “p-wave” ≥120 ms in DI, DII or DIII.
• Complete IAB or third grade blockage: defined as a biphasic “p wave” ≥120 ms in DII, DIII and AVF.
• IAB of second grade: an intermediate entity conformed by all thealterations previously described, which appear intermittently.
Recent studies using the criteria described above (3,5,7,8) reaffirmed IAB as a powerful supraventricular arrhythmias predictor. (5,7) However, not until now has anyone found association between cardioembolic strokes and the new criteria proposed by Bayes de Luna. In order to know if these new criteria are also safe and powerful predictors of cardioembolic stroke and be able to go ahead of the triggering cardiac pathology (atrial fibrillation) the study in this paper was performed.
Objective:
To establish IAB blockage as a new cerebrovascular risk factor prior to the onset of atrial fibrillation. In order to do this, study was designed in two phases:
• Phase 1 or “retrospective phase” planned to find an association between the IAB and Ischemic Strokes of patients with no evidence of cardioembolic etiology, situation classically included in the concept of Ischemic Strokes of Undetermined Source.
• Phase 2 or “prospective phase” with the objective to establish IAB as a cerebrovascular risk factor prior to the onset of supraventricular arrhythmias, as is atrial fibrillation.
Methodology of Phase 1 study:
Retrospective study of patients admitted to a General Hospital with the diagnosis at discharge of Ischemic Stroke (IS) of Undetermined Source (US), between January 2012 and December 2017, controlled by a group conformed by patients admitted in the same period of time for other reasons than Stroke or Arrhythmias, matched 1:1 by gender, age and cardiovascular risk factors (measure by
CHA 2 DS 2 -vasc scale).
During this time, a total of 356 patients were selected and divided into two groups:
• Group A (cases): 178 patients admitted by ischemic stroke (IS), that after etiologic studying were classified as having an undetermined source due to incomplete study (ISUCi), since none of them had transcranial eco doppler imaging tests (TED) and coagulopathy study. This classification was carried
out following the current TOAHS criteria for ischemic stroke (16).
• Group B (controls): made up of 178 patients admitted for other reasons (not stroke).
It is important to note that none of the 356 patients had a previous history of arrhythmias of any kind.
The methodology consisted in an analysis of admission ECG of all patientsincluded (cases and controls) by two different observers, looking for IAB according to the criteria proposed by Dr. Bayes de Luna in his consensus, defining the IAB of first grade or incomplete IAB (IABi) as a bimodal “p” ≥120ms located in leads I, II, or III, and complete IAB or third grade blockage (IABc) as a biphasic “p” wave
of ≥120ms in lower leads (II, III, and AVF).
Inclusion criteria: In the case group, patients were selected if after completing a serie of extensive studies, were labeled for ischemic stroke of undetermined source by incomplete study (ISUCi), following the current guidelines of the TOAHS classification for ischemic strokes (16) ; systematically ruling out Transient Ischemic Accidents (TIA), hemorrhagic stroke and ischemic stroke of any other etiology.
The extensive studies carried out and the exclusion criteria used for the case group were as follows:
• Computerized Tomography Scan (CTS): excluding those Lacunar stroke etiology (defined as ≤15mm in size) and hemorrhagic strokes.
• Doppler Ultrasound of Supra-Aortic Trunks (DSAT): excluding out obstructions >50% (cases of possible atherothrombotic etiology).
• Holter heart rate (during 24 hours): excluding those patients who during the test had detected supra-ventricular arrhythmias such as atrial fibrillation or atrial flutter of any duration.
• Transthoracic echocardiogram (TE): excluding cases in which intra chamber thrombi were detected or those patients with a ventricular ejection fraction <30%.
Due to limitations in our acute care unit, none of our patients underwent transcranial Doppler echo (TED) or coagulopathy study.
It is important to emphasize that in order to guarantee the greatest possible homogeneity of the population and thus be able to compare populations as similar as possible, strengthening the strength of independent association between groups. All the cases were matched 1 to 1 with the total sample of controls by age, gender and cardiovascular risk factors by means of the score presented by the CHA2DS2-vasc of each patient. That is to say that each case of an age, gender and specific CHA2DS2-vasc was paired with a control which presented these same characteristics. In this way we were able to correct the selection bias that we would have when comparing two heterogeneous samples (with different cardiovascular risk factors), thus increasing the strength of association of our results.
Finally, demographic, clinical, functional and cardiovascular risk factors measured through the CHA2DS2-vasc scale were analyzed in the total population. The data were obtained by reviewing all the documents of the clinical history related to this care process (including the database, the clinical course and complementary clinical tests in traditional format: medical and nursing evolution sheets, analytical, electrocardiograms and image studies).
Pathology Definition:
The functional status was assessed at admission using the Barthel score. Arterial Hypertension defined with systolic determination ≥140 mmHg, diastolic ≥90 mmHg or treatment with one or more hypotensive drug. Diabetes Mellitus I or II was defined by the presence of diagnosis in clinical history or by the presence of one or more hypoglycemic drugs in the prescription. Dyslipidemia was defined as a
total cholesterol ≥200 mg/dl, LDL ≥140 mg/dL, HDL ≤40 mg/dL, Triglycerides ≥150 mg/dL or a diagnosis present in clinical history or consumption of at least one drug lipid-lowering. The diagnoses of dementia, cognitive deterioration, peripheral arterial disease, heart failure and ischemic heart disease, as well as the active consumption of tobacco or alcohol were recovered directly from the clinical history.
Finally, the clinical classification of the stroke was made following the current classification criteria of Oxfordshire. (17)
Statistic Analysis:
The data was correlated and analyzed through the statistical program SPSS v.21. In comparison of averages, when the variables were continuous, Student’s t-test was used; for the comparison of dichotomous variables, the test used was X2. An unconditional logistic regression was performed to estimate the odds ratio (OR) and the 95% confidence intervals (CI) adjusted for possible confounding factors.
Results:
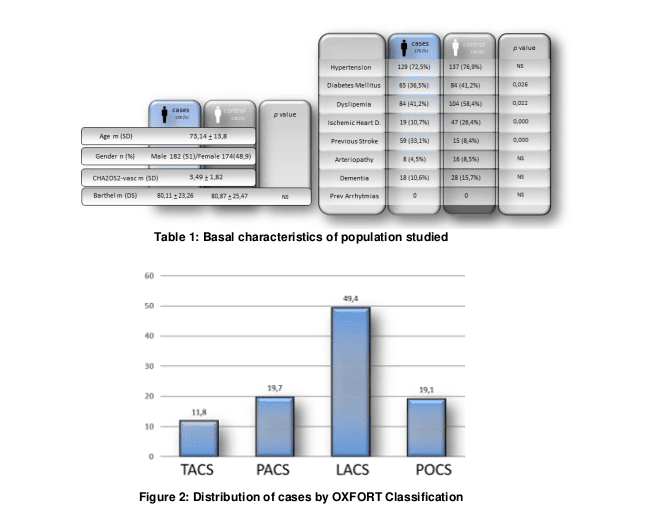
A total of 356 patients were included in the study, mean age of 73.1+13.8 years old, predominantly male (51%), with moderate functional dependence, measured through the Barthel index (80.49+24,36), distribute into two branches: Group A (cases) consisting of 178 patients with Ischemic stroke of undetermined source by incomplete study (ISUSi) and a Group B (controls) comprised of 178 patients admitted with out stroke. (Table I for the basal characteristics of population studied).
Both groups were matched 1:1 by age, gender and the total result of the cardiovascular risk scale CHA2DS2-vasc (mean 3.49+1.82), but not by each of their parameters individually. By analyzing cardiovascular risk factors separately no statistically significant differences were observed in baseline characteristics between both groups except for ischemic heart disease, Dyslipemia and Diabetes Mellitus, which were found to be more frequent in the control group (19% vs 47%, p 0.000), (41.2% vs. 58.4%, p 0.022) and (36.5 vs 41.2) respectively. (Table 1)
These results are most likely due to an inclusion bias, since all the controls included came from the hospital setting and not from the ambulatory setting (with higher comorbidity).
In describing the clinical characteristics of cases, we found a high proportion of lacunar-type events in 49.4% (LACS according to Oxfordshire classification), followed by those of posterior fossa or POCS and partial circulation or PACS (both with 19,1%) with less proportion of events of total circulation or TACS (11,8%) with a median Rankin scale at admission of 1,46+1,52. (Figure 2) The authors recognize a limitation due to the absence of Transcranial echo Doppler imaging studies, which would allow for more accurate exclusion of atherothrombotic events.
However, it is important to point out that 100% of the cases were carried out with Computerized Tomography Scan (CTS), Doppler Ultrasound of Supra-Aortic Trunks (DSAT), Holter of 24 hours, Transthoracic echocardiogram (TTE) and electrocardiographic records (EKG).
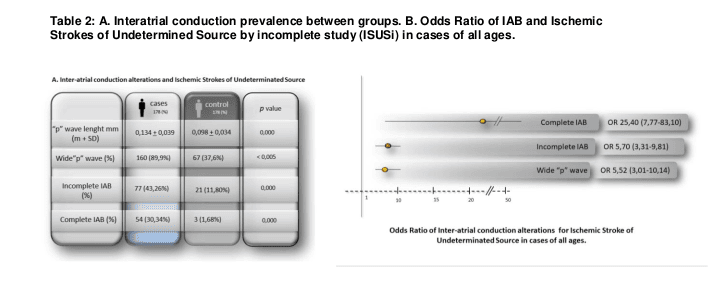
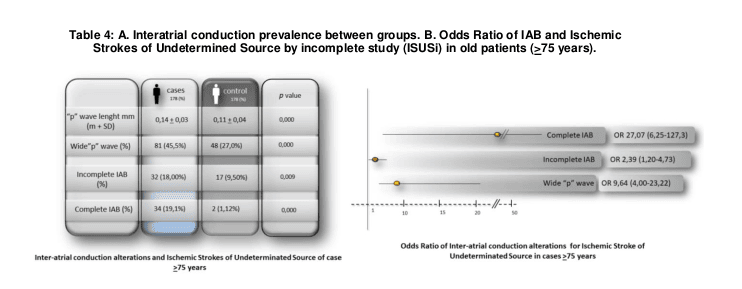
In Group A, patients with ISUSi presented a higher prevalence of interatrial conduction alterations compared to Group B (control), having a statistical significance in all of them: wide “p-wave” (89.9% vs 37.6% p 0.000), BIAi (43.26% vs. 11.8% p 0.000) and BIAc (30.34% vs. 1.68% p 0.000). (Table 2.A)
Table 2.B shows that the presence of a wide “p-wave” (WpW) (OR 5.52 3.01-10.14), the BIAi (OR 5.70 3.31-9.81) and the BIAc (OR 25,40 7,77-83,10) were significantly associated with the presence of ISUSi in general population; being the BIAc the most powerful of them all.
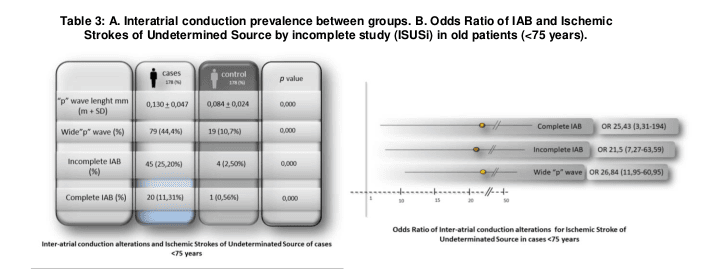
Nevertheless the most significant differences were found considering age (<75; ≥75), finding a stronger association WpW vs the new Inter arterial block criteria (IABi and IABc) in the younger group (OR 26,8(11,9-60,9) vs 21,5(7,27-63,5) and 25,43(3,3-194) while in contrast with a stronger association IABc and IABi vs WpW in the oldest group (OR 27,07(6,25-127,3) and 2,3(1,2-4,7) vs 9,6(4-23,2). (Table 3 and 4)
Discussion:
The cerebrovascular pathology is the third cause of death, the second cause of secondary dementia and the first cause of disability in the Western world (18) ; ischemic has been found to be eight times more frequent than hemorrhagic strokes and cardio embolic is considered the most common etiology of them all.
In this reality it is not surprising that efforts are directed toward finding alternatives that can address the consequences of this disease. Until now the successes in this field have been achieved by understanding the physiology of the problem, which has provided solutions in the acute and post-acute phases of stroke, correcting or at least minimizing consequences. However, despite all the advances in this field, stroke remains being a serious public health issue, therefore it’s time to look in a new direction establishing preventive measures before the cerebral vascular event.
It has been demonstrated that atrial fibrillation (AFib) is the main risk factor for the development of ischemic stroke of cardio-embolic etiology, a risk generated byalterations in atrial contractility and the blood stasis that it entails; However, it’s necessary to ask a simple question: does the cardio embolic risk begin with AFib or is it already present in phases prior to the clinical onset? In our study, in a population affected by ISUSi, we found a higher prevalence of interatrial conduction alterations with respect to a control group (without stroke).
Simple alterations easy to detect in a conventional EKG: wide “p-wave”, IABi and IABc that with statistical significance (p <0.000) were related to an ischemic stroke of undetermined source.
However, the most interesting results were found when analyzing the samples after dividing by age (<75; ≥75 years). We found that in the younger population (<75) the wide “p-wave” was the electrocardiographic anomaly related more importantly with the ISUCi group contrary to elderly population (≥75) in whom, for reasons that this study cannot clarify, the wide “p-wave” and the IABi lost strength of association with ISUSi group, being the IABc the anomaly with the most powerful
statistical association in elderlies with ISUSi.
These results can make us to consider that as years go by and diseases tends to accumulate (like hypertension, diabetes or heat failure) enlargement of atrials is produced, generating alterations in the normal EKG (as wider p-wave or partial interatrial conductions) debilitating its association with the occurrence of ISUSi.
Taking this into consideration, it could be that for elderlies the presence of complete IAB is necessary to consider, the interatrial blockage as a risk factor for stroke with a high sensitivity power.
The authors of this study recognize that since it is a retrospective study, of a limited population of patients, the results can be argumentative and more so in a population in which it was not possible to complete the etiologic study of strokes (such as the transcranial Doppler echo and the study of coagulopathy), however the results obtained are very encouraging.
The limitations of the study (previously described) make it impossible to postulate the atrial conduction alterations (p-wide, IABi and IABc) as predictors and new risk factor of cardio embolic stroke before the onset of AF; however, it raises the question of whether patients with ischemic stroke and disturbances in inter auricular conduction should be treated with anticoagulation before the IS and the FA.
Waiting for the second phase of this study, the results presented here could serve as a basis for new studies, more powerful, larger and methodologically better designed to clarify, if the IAB can be postulated as an economic, quick and simple tool that allow us to go years ahead of development of the stroke.
Conclusions:
After a statistical analysis of our population we can conclude that in our sample thelength of the p wave, the IABi and the IABc were related in a statistically significant way with the presence of the ISUCi. Specific score of cardiovascular risk (such as CHA2DS2-vasc) are of little use in the older population.
WPW and NIAB were significantly related to ISUCi. Relevant differences were found considering age, being more prevalent the presence of WPW in younger and IABc in elder. In particular the IABc not only showed the greatest statistical association, but they were shown as potentially useful risk factors to identify patients candidates to suffer an ischemic stroke.
The authors of the present study recognize the methodological limitations and the small number of participants. However, they consider that the results are sufficiently encouraging to carry out more studies that allow correcting on the limitations and amplifying the investigation.
If similar results are repeated in future studies, it could be hypothesized to alterations in inter atrial conduction, mainly to IABc, as a new cardiovascular risk factor that justified or not the possibility of performing thromboprophylaxis with anticoagulants prior not only to the appearance of the ischemic stroke, but even before the onset of supra ventricular arrhythmias type Atrial Fibrillation (AF).
Conflict of interests:
The authors of this work declare that they have no conflicts of interest.
Bibliografía:
- Willems JL, Robles de Medina EO, Bernard R, et al. Criteria for intraventricular conduction disturbances and pre-excitation. World Health Organization/International Society and Federation of Cardiology Task Force Ad hoc. J Am Coll Cardiol 1985;5:1261.
- Bachmann G. The inter-auricular time interval. Am J Physiol 1916;41:309.
- Diego Conde, Adrián Baranchuk. Bloqueo interauricular como sustrato anatómico-eléctrico de arritmias supraventriculares: síndrome de Bayés. Arch Cardiol Mex. 2014;84(1):32-40
- Bayés de Luna A, Cladellas M, Oter R, et al. Interatrial conduc- tion block and retrograde activation of the left atrium and paroxysmal supraventricular tachyarrhythmia. Eur Heart J. 1988;9:1112-8.
- GaryTse1*,EricTszHimLai1,JieMingYeo2 andBryanP.Yan. Electrophysiological Mechanisms of Bayés Syndrome: Insights from Clinical and Mouse Studies. Frontiers in Physiology. May 2016 | Volume 7
| Article 188. - Bayés de Luna A, X.Vinolas G, Martinez-Rubio A, et al. Third degree Inter-atrial block and supraventricular arrhythmias. Eu- ropace 1999; 1: 43-6.
- Lovely Chhabra* 1, Ramprakash Devadoss 1, Vinod K. Chaubey 1 and David H. Spodick. Interatrial Block in the Modern Era. Current Cardiology Reviews, 2014, 10, 181-189.
- Hideki Hayashi, MD, PhDn , Minoru Horie, MD, PhDn .Biphasic P wave in inferior leads and the development of atrial fibrillation. Journal of Arrhythmia 31 (2015) 376–380
- Vignendra Ariyarajah, Jaxon Fernandes , Mark Kranis , Sirin Apiyasawat , Kristin Mercado , David H. Spodick. Prospective evaluation of atrial tachyarrhythmias in patients with interatrial block. International Journal of Cardiology 118 (2007) 332–337
- Ariyarajah V, Fernandes J, Kranis M, et al. Prospective evalua- tion of atrial tachyarrhythmias in patients with interatrial block. Int J Cardiol. 2007;118:332-7.
- Ariyarajah V, Apiyasawat S, Najjar H, et al. Frequency of inter- atrial block in patients with sinus rhythm hospitalized for stroke and comparison to those without interatrial block. Am J Cardiol. 2007;99:49-52.
- Ariyarajah V, Puri P, Apiyasawat S, et al. Interatrial block: A novel risk factor for embolic stroke? Ann Noninvasive Electro- cardiol. 2007;12:15-20.
- Lorbar M, Levrault R, Phadke JG, Spodick DH. Interatrial block as a predictor of embolic stroke. Am J Cardiol 2005;95:667.
- Ariyarajah V, Puri P, Apiyasawat S, Spodick DH. Interatrial block: a novel risk factor for embolic stroke? Ann Noninvasive Electrocardiol 2007;12:15.
- Bayés de Luna A, Platonov P, Cosio FG, et al. Interatrial blocks. A separate entity from left atrial enlargement: A consensus report. J Electrocardiol. 2012;45:445-51.
- Teemu Vepsäläinen 1* , Markku Laakso2 , Seppo Lehto2, Auni Juutilainen1, Juhani Airaksinen1 and Tapani Rönnemaa. Prolonged P wave duration predicts stroke mortality among type 2 diabetic patients with prevalent non-major macrovascular disease. BMC Cardiovascular Disorders 2014,
14:168. - Bamford J, Sandercock P, Dennis M, Burn J, Warlow C. Classification and natural history of clinically identifiable subtypes of cerebral infarction. Lancet. 1991;337:1521–1526CrossRefPubMedGoogle Scholar
- Pedro Abizanda Soler, Leocardio Rodriguez Mañas. Tratado de Medicina Geriátrica. 2014;Capítulo 81;pagina 645. Enfermedad cerebro vascular en el anciano.
- Díaz-Guzmán J1, Egido-Herrero JA, Fuentes B, Fernández-Pérez C, Gabriel-Sánchez R, Barberà G, Abilleira S; Proyecto Ictus del Grupo de Estudio de Enfermedades Cerebrovasculares de la Sociedad Española de Neurología. Incidence of strokes in Spain: the Iberictus study. Data from
the pilot study. 2009 Jan 16-31;48(2):61-5. Bayés de Luna AJ. Bloqueo a nivel auricular. Rev Esp Cardiol. 1979;32:5. - Bayés de Luna A, Fort de Ribot R, Trilla E, et al. Electrocardio- graphicand vectorcardiographic study of interatrial conduction disturbances with left atrial retrograde activation. J Electro- cardiol. 1985;18:1-13.
- Jairath UC, Spodick DH. Exceptional prevalence of inter- atrial block in a general hospital population. ClinCardiol. 2001;24:548-50.
- Asad N, Spodick DH. Prevalence of interatrial block in a general hospital population. Am J Cardiol. 2003;91:609-10.a,
- Danai Kitkungvan, MD, ⁎ David H. Spodick, Interatrial block: is it time for more attention? Journal of Electrocardiology 42 (2009) 687–692
- Ariyarajah V, Asad N, Tandar A, Spodick DH. Interatrial block: pandemic
prevalence, significance, and diagnosis. Chest 2005;128:970. - Bayes de Luna A. Electrocardiographic alterations due to atrial pathology.
Clinical electrocardiography: a textbook. New York (NY): Futura Company; - p. 169.
- Larissa Fabritz, The power of P in the elderly: Small biphasic wave, big
impact. 2016 The Authors. Published by Elsevier Inc. on behalf of Heart
Rhythm Society - Simona Lattanzi a,⁎ , Claudia Cagnetti a , Alessandra Pulcini a , Marco Morelli b, Simone Maffei b, Leandro. Provinciali a , Mauro Silvestrini. The P-wave terminal force in embolic strokes of undetermined source. Journal of the Neurological Sciences 375 (2017) 175–178
- R.G. Hart, H.C. Diener, S.B. Coutts, Cryptogenic Stroke/ESUS International Working Group, et al., Embolic strokes of undetermined source: the case for a new clinical construct, Lancet Neurol. 13 (2014)
429–438. - R.J.Wityk,M.S.Pessin,R.F.Kaplan,etal.,Serialassessmentofacutestrokeusingthe NIH stroke scale, Stroke 25 (1994) 362–365.
- P.Davison,P.F.Clift,R.P.Steeds,Theroleofechocardiographyindiagnosis,monitor- ing closure and post-procedural assessment of patent foramen ovale, Eur. J. Echocardiogr. 11 (2010) i27–i34.
- J.Y. Lee, J.K. Song, J.M. Song, et al., Association between anatomic features of atrial septal abnormalities obtained by omni-plane transesophageal echocardiography and stroke recurrence in cryptogenic stroke patients with patent foramen ovale, Am. J. Cardiol. 106 (2010) 129–
134. - J.W.Kim,S.J.Kim,C.W.Yoon,etal.,Associationbetweentheamountofright-to- left shunt and infarct patterns in patients with cryptogenic embolic stroke: a transcrani- al Doppler study, Int. J. Stroke 8 (2013) 657–662.
- E.E. Williamson, J. Kirsch, P.A. Araoz, et al., ECG-gated cardiac CT angiography using 64-MDCT for detection of patent foramen ovale, AJR Am. J. Roentgenol. 190 (2008) 929–933.
- A.H. Katsanos, T. Psaltopoulou, T.N. Sergentanis, et al., Transcranial Doppler versus transthoracic echocardiography for the detection of patent foramen ovale in pa- tients with cryptogenic cerebral ischemia: a systematic review and diagnostic test accuracy meta-analysis, Ann. Neurol. 79 (2016) 625–635.36.
- Adams HP Jr., Bendixen BH, Kappelle LJ, et al. Classification of subtype
of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke,1993;24:35–41. - Baranchuk A, Villuendas R, Bayés Genis A, et al. Advanced interatrial block: A well defined electrocardiographic pat- tern with clinical arrhythmological implications. Europace. 2013;15:1822.
- Jurkko R, Mäntynen V, Tapanainen JM, et al. Non-invasive detection of conduction pathways to left atrium using mag- netocardiography: validation by intra-cardiac electroanatomic mapping. Europace. 2009;11:169-77.
- Baranchuk A, Michael K, Syed U, et al. Intermittent intera- trial block after electrical cardioversion for atrial fibrillation. J Electrocardiol. 2008;41:662-4.
- Pang H, Ronderos R, Pérez-Riera AR, et al. Reverse atrial elec- trical
remodeling: A systematic review. Cardiol J. 2011;18: 625-31.

